What We Do
DevPro Biopharma advances clinical development practices that enable molecules to become medicines faster, while lowering costs to generate value for patients and for our clients.
The DevPro Biopharma team covers all aspects of clinical development from study design through product submission and approval:
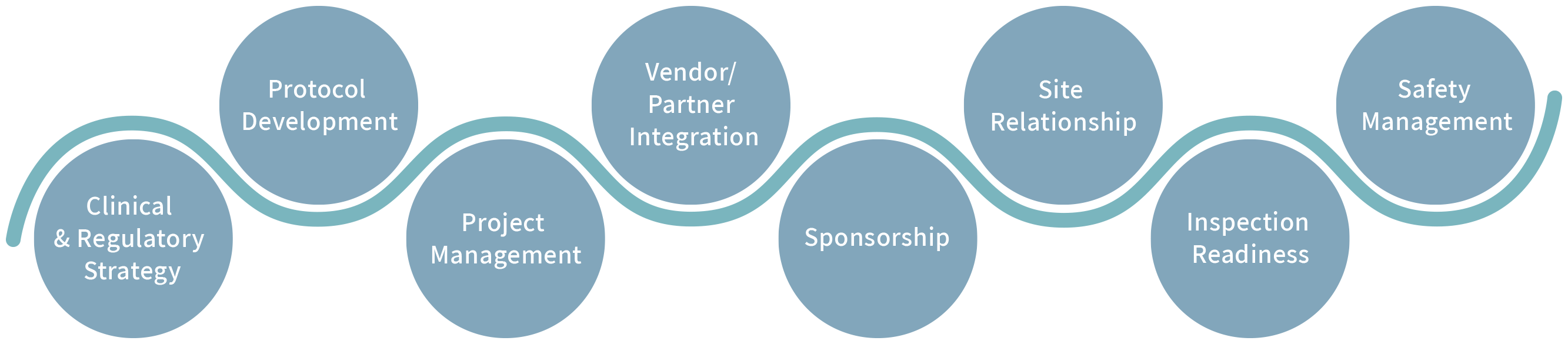
OUR EXPERTISE INCLUDES:
- Leading clinical programs and studies from protocol design through product approval, including clinical safety management, regulatory strategy, and inspection readiness
- Accelerating products to market, relative to competitors and standard pharmaceutical development timelines
- Managing both privately held and public organizations
- Navigating and excelling in a large, global pharmaceutical environment
- Global development and product approval across U.S., Europe, Asia, Latin America, as well as emerging markets, such as China
- Leading major regulatory sponsor inspections in U.S., EU, China, Japan
- International regulatory submissions and negotiation with regulatory bodies (FDA, EMA, PMDA, MHRA, Health Canada, TGA, NMPA, and more)
- Funding, building, and managing a start-up company
We bring development expertise and proven execution to help our clients design and execute programs that:
De-risk development path
We avoid technical design challenges such as optimizing primary endpoints, regulatory barriers such as inspection readiness, and executional issues such as critical- to-quality endpoint measures, as well as limiting the initial upfront development costs that increase an asset’s probability of success.

